Which Vitamin Can Be Produced by Intestinal Bacteria?
Introduction
The gut is continuously exposed both to toxic (e.chiliad., pathogenic microorganisms) and beneficial (e.g., dietary components, commensal bacteria) compounds and microorganisms; therefore, the intestinal immune system must maintain a good for you balance between active and suppressive immune responses. This balance is controlled not only past host allowed factors such as cytokines but also by a variety of environmental factors such as dietary components and the limerick of the commensal bacteria. Furthermore, several lines of evidence have demonstrated that allowed homeostasis in the intestine is related not but to abdominal health but besides to the health of the whole body (one–3). Therefore, immune regulation by ecology factors is attracting attention as a ways of maintaining immunological health and preventing many diseases.
Nutrients are essential for the development, maintenance, and function of the host immune system (4, 5). Vitamins are essential micronutrients that are synthesized by bacteria, yeasts, and plants, but not by mammals. Therefore, mammals must obtain vitamins from the nutrition or rely on their synthesis by commensal bacteria in the gastrointestinal tract. Some vitamins are water-soluble (east.k., vitamin B family unit and vitamin C), whereas others are fat-soluble (east.thou., vitamins A, D, Eastward, and K). Water-soluble vitamins are not stored by the body and any excess is excreted in the urine; therefore, it is important to consume a diet containing the necessary amounts of these vitamins. Vitamin deficiency associated with insufficient dietary intake occurs not only in developing countries but also in developed countries as a upshot of increased use of unbalanced diet (half dozen).
In addition to the nutrition, the commensal bacteria are recognized as important players in the control of host health (7–nine). From the point of view of vitamins, commensal bacteria are both providers and consumers of B vitamins and vitamin K. Although dietary B vitamins are generally captivated through the small intestine, bacterial B vitamins are produced and absorbed mainly through the colon (ten, 11), indicating that dietary and gut microbiota-derived B vitamins are peradventure handled differently by the human body. B vitamins are important cofactors and coenzymes in several metabolic pathways, and information technology has been reported recently that B vitamins too play important roles in the maintenance of allowed homeostasis (12, thirteen). Thus, both dietary components and the gut microbiota modulate host immune role via B vitamins. Hither, nosotros review the metabolism and function of dietary and gut microbiota-derived B vitamins in the control of host immunity.
Vitamin B1
Vitamin B1 (thiamine) is a cofactor for several enzymes, including pyruvate dehydrogenase and α-ketoglutarate dehydrogenase, which are both involved in the tricarboxylic acrid (TCA) cycle (xiv, 15). Earth Health Organization (WHO)/Food and Agriculture Organization (FAO) recommend a daily vitamin B1 intake of 1.1–one.ii mg for adult (16). Vitamin B1 deficiency causes sluggishness and, if left untreated, can develop into beriberi, a illness that affects the peripheral nervous system and cardiovascular system. Accumulating evidence suggests that energy metabolism—specially the balance betwixt glycolysis and the TCA wheel—is associated with the functional command of immune cells, in what is now referred to as immunometabolism (17).
Immunometabolism is well studied in T cells and macrophages; quiescent or regulatory-type cells (e.m., naive T cells, Treg cells, and M2 macrophages) use the TCA bike for energy generation, whereas activated or pro-inflammatory cells (e.yard., Th1, Th2, Th17, and M1 macrophages) employ glycolysis (18, 19).
Previously, we examined B cell immunometabolism in the intestine. In the intestine, naïve immunoglobulin (Ig) Grand+ B cells differentiate into IgA+ B cells in Peyer's patches (PPs) by course switching, and and so IgA+ B cells differentiate into IgA-producing plasma cells in the intestinal lamina propria (xx). Naïve B cells in PPs preferentially use a vitamin B1-dependent TCA bicycle for the generation of ATP. However, in one case the B cells differentiate into IgA-producing plasma cells, they switch to using glycolysis for the generation of ATP and shift to a catabolic pathway for the production of IgA antibody (Effigy 1). Consistent with the importance of vitamin B1 in the maintenance of the TCA bicycle, mice fed a vitamin B1-deficient diet show dumb maintenance of naïve B cells in PPs, with lilliputian issue on IgA-producing plasma cells. Since PPs are the primary sites of induction of antigen-specific IgA responses, PP regression induced past vitamin B1 deficiency leads to decreased IgA antibiotic responses to oral vaccines (21).

Figure 1. Vitamin B1 and B2-mediated immunometabolism in B cell differentiation in the intestine. Vitamin B1 acts as a cofactor for enzymes such as pyruvate dehydrogenase and α-ketoglutarate dehydrogenase that are involved in the TCA wheel. Vitamin B2 acts every bit a cofactor for enzymes such as succinate dehydrogenase in the TCA cycle and acyl-CoA dehydrogenase in fatty acid oxidation (FAO, also known equally β-oxidation). Naïve B cells preferentially use the TCA cycle for efficient energy generation. Once B cells are activated to differentiate into IgA-producing plasma cells, they utilize glycolysis for the production of IgA antibody.
Vitamin B1 is plant in high concentrations as thiamine pyrophosphate (TPP) in meat (peculiarly pork and chicken); eggs; cereal sprouts and rice bran; and beans. Dietary TPP is hydrolyzed past alkaline phosphatase and converted to complimentary thiamine in the modest intestine (22). Gratuitous thiamine is absorbed by the intestinal epithelium via thiamine transporters (e.g., THTR-1, THTR-ii) and transported to the claret for distribution throughout the torso (eleven). Free thiamine is converted back to TPP and is used for energy metabolism in the TCA cycle.
Various types of intestinal bacteria, mostly in the colon, also produce vitamin B1 as both free thiamine and TPP (11, 23). In the colon, free bacterial thiamine is captivated mainly by thiamine transporters, transported to the claret, and distributed throughout the torso; this machinery is similar to how free dietary thiamine is taken upwards in the small intestine. However, unlike in the modest intestine, TPP produced past the gut microbiota is not converted to free thiamine, considering alkaline phosphatase is not secreted in the colon (24). Instead, TPP is absorbed direct by the colon via TPP transporters (e.g., TPPT-1) that are highly expressed on the apical membrane of the colon (25). The captivated TPP enters the mitochondria via MTPP-i, a TPP transporter that is expressed in the mitochondrial inner membrane and is used equally a cofactor for ATP generation (26). This suggests that bacterial TPP is important for energy generation in the colon. Thus, dietary and bacterial vitamin B1 appears to have different roles in the host.
The vitamin B1 construction consists of a thiazole moiety linked to a pyrimidine moiety. Bacteria obtain the thiazole moiety from glycine or tyrosine and 1-deoxy-d-xylulose-5-phosphate, and plants and yeasts synthesize it from glycine and 2-pentulose (27–30). In both bacteria and plants, the pyrimidine moiety is derived from 5-aminoimidazole ribonucleotide, an intermediate in the purine pathway (29). Metagenomic analyses of the homo gut microbiota predict that Bacteroides fragilis and Prevotella copri (phylum Bacteroidetes); Clostridium difficile, some Lactobacillus spp., and Ruminococcus lactaris (Firmicutes); Bifidobacterium spp. (Actinobacteria); and Fusobacterium varium are vitamin B1 producers (Tabular array 1) (10, 46), implying that many intestinal leaner possess a complete vitamin B1 synthesis pathway, which includes pathways for the synthesis of thiazole and pyrimidine. Indeed, Lactobacillus casei produces thiamine during the production of fermented milk drinks (31), and Bifidobacterium infantis and B. bifidum produce thiamine in civilization supernatant (32). However, Faecalibacterium spp. (Firmicutes) lack a vitamin B1 synthesis pathway even though they require vitamin B1 for their growth (10). Therefore, these leaner must obtain their vitamin B1 from other bacteria or from the host diet via a thiamine transporter, suggesting that there is competition for vitamin B1 between the host and certain abdominal leaner.

Table 1. Vitamin B family producing leaner.
Vitamin B2
Vitamin B2 (riboflavin) and its active forms (flavin adenine dinucleotide [FAD] and flavin mononucleotide [FMN]) are cofactors for enzymatic reactions in the TCA bike and in fatty acid oxidization (also known as β-oxidization) (fifteen). WHO/FAO recommends a daily vitamin B2 intake of i.0–1.three mg for adults (16). Vitamin B2 deficiency suppresses the activity of acyl-CoA dehydrogenases involved in the oxidation of fat acids to generate acetyl-CoA, which is used by mitochondria to produce ATP via the TCA cycle. Fatty acid oxidization is involved in the activation, differentiation, and proliferation of allowed cells through the generation of acetyl-CoA and its entry into TCA cycle (47). This suggests that vitamin B2 is associated with the control of differentiation and function of immune cells through regulation of fat acid oxidization (Effigy 1); nonetheless, the immunological roles of vitamin B2 in the control of host amnesty remain to be investigated. In addition to energy generation, vitamin B2 is associated with reactive oxygen species (ROS) generation in immune cells through the priming of NADPH oxidase two (48); ROS are important effector and signaling molecules in inflammation and amnesty.
Vitamin B2 is establish at loftier levels in leafy green vegetables, liver, and eggs. Dietary vitamin B2 exists equally FAD or FMN and is converted to free riboflavin by FAD pyrophosphatase and FMN phosphatase in the small intestine (49, l). Free riboflavin is captivated via riboflavin transporter expressed on the epithelium of the pocket-size intestine and is then released into the claret. In the blood, gratis riboflavin is converted back to FAD or FMN and distributed throughout the trunk (51–53).
Bacterial vitamin B2 is synthesized from guanosine triphosphate (GTP) and d-ribulose 5-phosphate (54). Bacterial vitamin B2 exists as free riboflavin, which is direct absorbed in the large intestine, converted to FAD or FMN, and distributed throughout the body as described above (23). A metagenome analysis of the human gut microbiota past Magnúsdóttir et al. (10) has predicted that Bacteroides fragilis and Prevotella copri (Bacteroidetes); Clostridium difficile, Lactobacillus plantarum, Fifty. fermentum, and Ruminococcus lactaris (Firmicutes) express factors essential for vitamin B2 synthesis, suggesting that these leaner are an of import source of vitamin B2 in the large intestine (Table 1). In contrast, Bifidobacterium spp., and Collinsella spp. (Actinobacteria) lack a vitamin B2 pathway. Supplementation of fermented soymilk containing Lactobacillus plantarum with riboflavin deficient nutrition has been shown to promote vitamin B2 production and forbid vitamin B2 deficiency in mice (35). L. fermentum isolated from sourdough can synthesize riboflavin in vitro (36). Furthermore, contempo evidence indicates that some species in Bacteroidetes phylum produce more riboflavin than practice Actinobacteria and Firmicutes phyla (55). Nonetheless, Actinobacteria and Firmicutes phyla notwithstanding express riboflavin transporter and the enzymes necessary for FAD and FMN generation from gratuitous riboflavin (i.e., FAD synthases and flavin kinases) (ten, 56), suggesting that all bacteria, including those that are unable to synthesize vitamin B2 themselves, require FAD and FMN for their growth and survival. Thus, every bit with vitamin B1, at that place is probable competition for riboflavin between the host and the commensal leaner.
In addition to being able to produce vitamin B2, some bacteria (e.chiliad., commensals such as Lactobacillus acidophilus and pathogens such as Mycobacterium tuberculosis and Salmonella typhimurium) produce the vitamin B2 intermediate (57–59), 6-hydroxymethyl-viii-d-ribityllumazine (60, 61). 6-Hydroxymethyl-eight-d-ribityllumazine binds to major histocompatibility complex class I-related gene protein (MR1) on antigen-presenting cells; this causes mucosal-associated invariant T (MAIT) cells, an abundant population of innate-like T cells, to produce cytokines such equally interferon gamma and interleukin (IL) 17, which contribute to host defense confronting pathogens (Figure ii) (62). It is thought that stimulation by commensal bacteria contributes to the development and activation of MAIT cells for immunological surveillance against pathogens. MAIT cells as well produce inflammatory cytokines and have tissue-homing properties, suggesting that these cells are likewise involved in the development of autoimmune and inflammatory diseases (63).
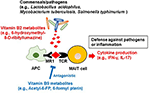
Figure ii. Regulation of MAIT cells by microbial metabolites derived from vitamin B2 and B9. Commensal leaner/pathogens produce the vitamin B2 metabolite 6-hydroxymethyl-eight-D-ribityllumazine. It binds to major histocompatibility circuitous (MHC) related poly peptide (MR1) on antigen-presenting cells, which activate mucosal associated invariant T (MAIT) cells to promote the production of inflammatory cytokines such as IFN-γ and IL-17. These reactions contribute to defense force against pathogens and conversely are associated with inflammation. In contrast, the vitamin B9 metabolite acetyl-half dozen-formylpterin binds as an antagonist to MR1, thus inhibiting the activation of MAIT cells.
Vitamin B3
Vitamin B3 (niacin) is generally known as nicotinic acid and nicotinamide. These compounds are precursors of nicotinamide adenine dinucleotide (NAD), a coenzyme in the cellular redox reaction with a central role in aerobic respiration. WHO/FAO recommends a daily vitamin B3 intake of 11–12 mg for adults (16).
Vitamin B3 is likewise a ligand of GPR109a, a M-protein coupled receptor expressed on several types of cells, including allowed cells (64). Vitamin B3–GPR109a signaling induces differentiation of regulatory T cells and suppresses colitis in a GPR109a-dependent fashion (65). Vitamin B3 also inhibits the production of the pro-inflammatory cytokines IL-1, IL-6, and tumor necrosis factor alpha (TNF-α) past macrophages and monocytes (Figure 3) (66). Thus, vitamin B3 has anti-inflammatory properties by modulating host allowed cells and playing an important part in the maintenance of immunological homeostasis. Indeed, in humans, vitamin B3 deficiency causes pellagra, which is a disease characterized by intestinal inflammation, diarrhea, dermatitis, and dementia (67).
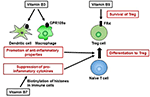
Figure 3. Pivotal roles of vitamins B3, B7, and B9 in maintenance of immunological homeostasis. Vitamin B3 binds to GPR109a in dendritic cells and macrophages, and GPR109a signaling leads to an increase in anti-inflammatory backdrop, resulting in differentiation into regulatory T cells (Treg). Vitamin B7 binds to histones and, past histone biotinylation, suppresses the secretion of pro-inflammatory cytokines. Once naïve T cells differentiate into Treg cells, they highly express folate receptor iv (FR4). Consistent with this finding, vitamin B9 is required for the survival of Treg cells.
Unlike the other B vitamins, vitamin B3 can be generated past mammals via an endogenous enzymatic pathway from tryptophan and is stored in the liver, although information technology is also obtained from the diet (68). Animate being-based foods such as fish and meat contain vitamin B3 as nicotinamide, and plant-based foods such every bit beans contain vitamin B3 as nicotinic acid. Both nicotinamide and nicotinic acrid are directly absorbed through the small intestine, where nicotinic acid is converted to nicotinamide.
Vitamin B3 is also synthesized from tryptophan past intestinal bacteria (69, lxx). Bacteroides fragilis and Prevotella copri (Bacteroidetes); Ruminococcus lactaris, Clostridium difficile (Firmicutes); Bifidobacterium infantis (Actinobacteria); Helicobacter pylori (Proteobacteria); and Fusobacterium varium (Fusobacteria) possess a vitamin B3 biosynthesis pathway (Tabular array 1) (10, 71). Thus, many abdominal bacteria from various genera can produce vitamin B3, suggesting that both dietary and commensal leaner-derived vitamin B3 are important for host immunity.
Vitamin B5
Vitamin B5 (pantothenic acid) is a precursor of coenzyme A (CoA), which is an essential cofactor for the TCA cycle and fat acid oxidation (72). WHO/FAO recommends a daily vitamin B5 intake of 5.0 mg for adults (16). Similar vitamins B1 and B2, vitamin B5 is involved in the control of host immunity via free energy generation by immune cells. Vitamin B5 deficiency causes allowed diseases such equally dermatitis, besides as not-immune-related weather such every bit fatigue and insomnia (73). In a randomized, double-blind, placebo-controlled written report in adults, dietary supplementation with vitamin B5 was shown to improve facial acne (74), suggesting that epithelial bulwark function improves via the promotion of keratinocyte proliferation and differentiation into fibroblasts (75). To maintain vitamin B5 levels during times of deficiency, CoA is converted back to vitamin B5 or cysteamine via pantetheine (76). Notwithstanding, cysteamine inhibits peroxisome proliferator-activated receptor gamma (PPARγ) signaling, causing inflammation (77). Indeed, colitis has been improved in pantetheine-producing-enzyme knockout mice (78). Thus, vitamin B5 deficiency causes inflammation through both dysfunction of the epithelial bulwark and the production of pro-inflammatory molecules.
In terms of immune responses, vitamin B5 enhances protective action against Mycobacterium tuberculosis infection by promoting innate immunity and adaptive immunity. In mice, vitamin B5 supplementation activates phagocytosis and cytokine production (including IL-6 and TNF-α) by macrophages and subsequently promotes Th1 and Th17 responses for the clearance of M. tuberculosis from the lungs (79). Thus, vitamin B5 contributes to host defence by activating immune responses, suggesting that this vitamin has an of import role every bit a natural adjuvant.
Vitamin B5 is plant in high concentrations every bit CoA or phosphopantetheine in liver, eggs, craven, and fermented soybeans. CoA and phosphopantetheine are converted to gratuitous pantothenic acid by endogenous enzymes such equally phosphatase and pantetheinase in the small intestine. Free pantothenic acrid is captivated via sodium-dependent multivitamin transporter (SMVT) expressed on the epithelium of the small intestine and is then released into the claret (80). Finally, costless pantothenic acrid is converted back to CoA and distributed throughout the trunk, especially to the liver and kidney.
Bacterial vitamin B5 is synthesized from 2-dihydropantoate and β-alanine via de novo synthesis pathways (10). Bacterial vitamin B5 exists as gratuitous pantothenic acid, which is straight absorbed in the big intestine, converted to CoA, and distributed in the same style equally dietary vitamin B5. According to a genomic assay, Bacteroides fragilis and Prevotella copri (Bacteroidetes); some Ruminococcus spp. (R. lactaris and R. torques) (Firmicutes); Salmonella enterica and Helicobacter pylori (Proteobacteria) possess a vitamin B5 biosynthesis pathway, indicating that abdominal commensal bacteria can produce vitamin B5. In contrast, most Fusobacterium (Fusobacteria) and Bifidobacterium spp. (Actinobacteria) and some strains of Clostridium difficile, Faecalibacterium spp., and Lactobacillus spp. (Firmicutes) lack such a pathway (Tabular array one), although some of them practise express pantothenic acrid transporter to employ vitamin B5 for free energy generation (10), suggesting that these bacteria compete with the host for vitamin B5.
Vitamin B6
Vitamin B6 exists in several forms, including as pyridoxine, pyridoxal, and pyridoxamine. These forms of vitamin B6 are precursors of the coenzymes pyridoxal phosphate (PLP) and pyridoxamine phosphate (PMP), which are involved in a multifariousness of cellular metabolic processes, including amino acid, lipid, and carbohydrate metabolism (81). WHO/FAO recommends a daily vitamin B6 intake of 1.3–ane.7 mg for adults (xvi). Vitamin B6 deficiency is associated with the development of inflammatory diseases such as allergy and rheumatoid arthritis, as well as with neuronal dysfunction (82–84). Vitamin B6 deficiency disrupts the Th1–Th2 balance toward an excessive Th2 response, resulting in allergy (85). Moreover, low plasma vitamin B6 levels, together with increased levels of pro-inflammatory cytokines such as TNF-α and IL-six, have been observed in patients with rheumatoid arthritis (86). Notwithstanding, the mechanism underlying the regulation of inflammation by vitamin B6 is currently unknown. Vitamin B6 contributes to intestinal allowed regulation through the metabolism of the lipid mediator sphingosine 1-phosphate (S1P). S1P regulates lymphocyte trafficking into the intestines, specially in the big intestine. Lymphocyte trafficking is dependent on S1P slope which is created past S1P production and its degradation. S1P deposition is mediated past S1P lyase that requires vitamin B6 as a cofactor. The administration of vitamin B6 adversary impairs S1P lyase activity and creates an inappropriate S1P gradient, resulted in impairing lymphocyte migration from lymphoid tissues and reducing the numbers of lymphocytes in the intestines (87). The lymphocytes located betwixt gut epithelial cells are known as intraepithelial cells (IELs) which are involved in the protection confronting pathogens (88). Therefore, vitamin B6 is important role for immunosurveillance in the intestines.
Vitamin B6 is arable in fish, chicken, tofu, sweetness potato, and avocado. Dietary vitamin B6 exists as PLP or PMP; it is converted to free vitamin B6 past endogenous enzymes such as pyridoxal phosphatase and is so absorbed past the small-scale intestine. Although absorption of vitamin B6 through acidic pH-dependent and carrier-mediated ship has been shown, an intestinal pyridoxine transporter is even so to be identified in any mammalian species (11). After the absorption of costless vitamin B6, it enters the blood and is converted back to PLP or PMP.
Microbial vitamin B6 is synthesized as PLP from deoxyxylulose 5-phosphate and 4-phosphohydroxy-Fifty-threonine or from glyceraldehyde-3-phosphate and d-ribulose v-phosphate (10). In the large intestine, bacteria-derived PLP is converted to free vitamin B6, which is absorbed past passive transport, transported to the claret, and distributed throughout the body.
Metagenomic analysis has shown that Bacteroides fragilis and Prevotella copri (Bacteroidetes), Bifidobacterium longum and, Collinsella aerofaciens (Actinobacteria), and Helicobacter pylori (Proteobacteria) possess a vitamin B6 biosynthesis pathway. Bacteroidetes and Proteobacteria likely produce vitamin B6 starting from deoxyxylulose 5-phosphate and four-phosphohydroxy-l-threonine, whereas Actinobacteria likely commencement from glyceraldehyde-iii-phosphate and d-ribulose 5-phosphate. In contrast, most Firmicutes genera (Veillonella, Ruminococcus, Faecalibacterium, and Lactobacillus spp.), except for some Clostridium (C. bartlettii, C. leptum, C. methylpentosum, and C. sporogenes) and Lactobacillus spp. (L. brevis and 50. ruminis) lack a vitamin B6 biosynthesis pathway (x) (Table i).
Vitamin B7
Vitamin B7 (biotin) is a cofactor for several carboxylases that are essential for glucose, amino acid, and fatty acid metabolism (89). For instance, vitamin B7 is an essential cofactor for acetyl-CoA carboxylase and fatty acid synthase, which are enzymes involved in fatty acrid biosynthesis (90, 91). Thus, vitamin B7 likely influences immunometabolism. WHO/FAO recommends a daily vitamin B7 intake of 30 μg for adults (sixteen). Vitamin B7 suppresses gene expression by binding to (biotinylating) histones; these genes include that encoding NF-κB, which is a major signaling molecule for the product of several pro-inflammatory cytokines (east.g., tumor necrosis factor alpha, IL-1, IL-half-dozen, IL-8) (92, 93). Nuclear transcription of NF-κB is activated in response to vitamin B7 deficiency (94), suggesting that biotinylation of histones suppresses the expression of genes encoding pro-inflammatory cytokines in NF-κB signaling (Figure 3). Therefore, vitamin B7 has anti-inflammatory effects past inhibiting NF-κB activation, and dietary vitamin B7 deficiency causes inflammatory responses via enhanced secretion of pro-inflammatory cytokines (95, 96).
Vitamin B7 is abundant in foods such as nuts, beans, and oilseed. However, raw egg-white contains a large amount of avidin, which binds strongly to vitamin B7 and prevents its absorption in the gut (97). Therefore, vitamin B7 deficiency tin can be caused not only by insufficient vitamin B7 intake, but too by excessive intake of raw egg-white. Dietary biotin exists as a complimentary protein-bound course or as biocytin (11). In the small intestine, biotinidase releases gratis biotin from the jump protein and the gratuitous biotin is absorbed via the biotin transporter SMVT (98).
Vitamin B7 is besides produced by abdominal bacteria as gratuitous biotin synthesized from malonyl CoA or pimelate via pimeloyl-CoA (99, 100). Bacterial gratis biotin is absorbed by SMVT expressed in the colon (23, 101). Metagenomic analysis has shown that Bacteroides fragilis and Prevotella copri (Bacteroidetes); Fusobacterium varium (Fusobacteria) and Campylobacter coli (Proteobacteria) possess a vitamin B7 biosynthesis pathway (ten). In dissimilarity, Prevotella spp. (Bacteroidetes), Bifidobacterium spp. (Actinobacteria), and Clostridium, Ruminococcus, Faecalibacterium, and Lactobacillus spp. (Firmicutes) lack such a pathway (Table 1); however, they do limited free biotin transporter (10, 102), suggesting that these bacteria also use dietary and bacterial vitamin B7 and therefore may compete with the host. Thus, free biotin may influence the limerick of the gut microbiota, because biotin is necessary for the growth and survival of the microbiota. Indeed, biotin deficiency leads to gut dysbiosis and the overgrowth of Lactobacillus murinus, leading to the development of alopecia (103). Furthermore, vitamin B7 product appears to continue in a cooperative way amongst dissimilar intestinal leaner; Bifidobacterium longum in the intestine produces pimelate, which is a precursor of vitamin B7 that enhances vitamin B7 production by other intestinal leaner (104).
Vitamin B9
Vitamin B9 (folate), in its active grade equally tetrahydrofolate, is a cofactor in several metabolic reactions, including DNA and amino acid synthesis. WHO/FAO recommends a daily vitamin B9 intake of 400 μg for adults (xvi). Owing to the loftier requirement of vitamin B9 by red blood cells, vitamin B9 deficiency leads to megaloblastic anemia (23). Vitamin B9 deficiency also inhibits the proliferation of human being CD8+ T cells in vitro by absorbing the cell cycle in the S phase and increasing the frequency of Deoxyribonucleic acid damage (105). Moreover, vitamin B9 contributes to the maintenance of immunologic homeostasis. Regulatory T cells (Treg) express loftier levels of vitamin B9 receptor (folate receptor iv [FR4]). Administration of anti-FR4 antibiotic results in specific reduction in the Treg prison cell population (106), suggesting that the vitamin B9–FR4 axis is required for Treg cell maintenance. In vitro culture of Treg cells nether vitamin B9-reduced weather leads to dumb jail cell survival, with decreased expression of anti-apoptotic Bcl2 molecules, although naïve T cells retain the ability to differentiate into Treg cells; this suggests that vitamin B9 is a survival cistron for Treg cells (87). Consistent with these findings, deficiency of dietary vitamin B9 results in reduction of the Treg cell population in the modest intestine (107, 108). Since Treg cells play an of import role in the prevention of excessive immune responses (109), mice fed a vitamin B9-deficient diet exhibit increased susceptibility to abdominal inflammation (107).
Foods such every bit beefiness liver, greenish leafy vegetables, and asparagus contain high levels of vitamin B9. Vitamin B9 exists every bit both mono- and polyglutamate folate species in the diet (110). Folate polyglutamate is deconjugated to the monoglutamate form so captivated in the small intestine via folate transporters such as proton-coupled folate transporter (PCFT) (eleven, 111). In the intestinal epithelium, folate monoglutamate is converted to tetrahydrofolate (THF), an active grade and cofactor, before being transported to the claret (111).
Intestinal leaner synthesize vitamin B9 equally THF from GTP, erythrose 4-phosphate, and phosphoenolpyruvate (38, 112). Bacterial THF is straight captivated in the colon via PCFT and distributed through the body by the blood (113). Metagenomic analysis has shown that Bacteroides fragilis and Prevotella copri (Bacteroidetes); Clostridium difficile, Lactobacillus plantarum, 50. reuteri, L. delbrueckii ssp. bulgaricus, and Streptococcus thermophilus (Firmicutes), some species in Bifidobacterium spp (Actinobacteria); Fusobacterium varium (Fusobacteria) and Salmonella enterica (Proteobacteria) possess a folate biosynthesis pathway (Table 1) (10, 40). This suggests that almost all species in all phyla produce folate. Indeed, dietary supplementation with Bifidobacterium probiotic strains (B. adolescentis and B. pseudocatenulatum) enhances folate production in the large intestine of folate-scarce rats and folate-gratis culture medium (38, 41, 114). Furthermore, Lactobacillus plantarum, L. delbrueckii ssp. bulgaricus, and L. reuteri enhance folate product in bacterial culture supernatant lacking the components needed for folate synthesis (38, 39, 115).
In commensal bacteria, a vitamin B9 metabolite, six-formylpterin (6-FP), is produced by photodegradation of folic acid (116). Like the vitamin B2 metabolite 6-hydroxymethyl-8-d-ribityllumazine, six-FP binds to MR1, but dissimilar vi-hydroxymethyl-8-d-ribityllumazine information technology cannot actuate MAIT cells (62, 117). An analog of 6-FP, acetyl-half dozen-FP, is an antagonist of MR1, which inhibits MAIT jail cell activation (118). Equally mentioned in the section on vitamin B2, 6-hydroxymethyl-8-d-ribityllumazine activates MAIT cells, which provide defense against pathogens, so vitamin B9 metabolites may suppress excess MAIT prison cell responses and prevent excessive allergic and inflammatory responses (Figure ii). The quantitative balance betwixt dietary vitamin B2 and B9 and the limerick of the microbiota and its ability to metabolize these vitamins may be keys to understanding MAIT-prison cell-mediated homeostasis in the intestine.
Vitamin B12
Vitamin B12 (cobalamin) is a cobalt-containing vitamin that, in its active forms of methylcobalamin and adenosylcobalamin, catalyzes methionine synthesis (119). WHO/FAO recommends a daily vitamin B12 intake of ii.4 μg for adults (16). Together with vitamin B6 and B9, vitamin B12 plays important roles in red claret prison cell formation and nucleic acid synthesis, especially in neurons. Therefore, vitamin B12 deficiency causes megaloblastic anemia and nervous organization symptoms such equally numbness of the hands and anxiety (119). In terms of host immunity, dietary vitamin B12 deficiency decreases the number of CD8+ T cells and suppresses natural killer T-prison cell action in mice; supplementation with methylcobalamin improves these conditions (120), suggesting that vitamin B12 contributes to the immune response via CD8+ T cells and natural killer T cells.
Beef liver, bivalves, fish, craven, and eggs contain high levels of vitamin B12. Dietary vitamin B12 exists in circuitous with dietary protein and is decomposed to complimentary vitamin B12 by pepsin in the stomach. Free vitamin B12 is absorbed by the epithelial cells of the small intestine via intrinsic factor (IF), a gastric glycoprotein. Within the epithelial cells, IF-vitamin B12 complex is decomposed to free vitamin B12 by lysosome and so released into the blood, where it is converted to the active form and distributed throughout the body (121, 122).
Bacterial vitamin B12 is synthesized from precorrin-2 to produce adenosylcobalamin (x), which is absorbed direct by the big intestine and distributed throughout the torso; the machinery underlying this absorption is currently unclear. Metagenomic assay has predicted that Bacteroides fragilis and Prevotella copri (Bacteroidetes); Clostridium difficile, Faecalibacterium prausnitzii and Ruminococcus lactaris (Firmicutes); Bifidobacterium animalis, B.infantis, and B.longum (Actinobacteria); Fusobacterium varium (Fusobacteria) possess a vitamin B12 biosynthesis pathway (Table 1) (10, 32, 42, 43, 45). Indeed, Lactobacillus plantarum and Fifty. coryniformis isolated from fermented food produce vitamin B12 (33), and Bifidobacterium animalis synthesizes vitamin B12 during milk fermentation (123).
Conclusion
B-vitamin-mediated immunological regulation is specific to dissimilar immune cells and allowed responses: that is, different B vitamins are required for different immune responses (Figure 4). Information technology was once thought that B vitamins were obtained merely from the diet; however, nosotros know now that this is not the case and that the intestinal microbiota is likewise an important source of vitamins. Inside the abdominal microbiota, not all bacteria produce B vitamins and some bacteria utilize dietary B vitamins or B vitamins produced by other intestinal bacteria for their own needs; therefore, at that place may be competition between the host and the abdominal microbiota for B vitamins (Figure four). Inquiry in this field is complicated, because non only does the composition of the intestinal microbiota vary amongst individuals, simply also the composition of the diet tin alter both the composition and function of the intestinal microbiota. Therefore, vitamin-mediated immunological maintenance also varies amongst individuals. Further examinations in this field are needed, and the new information uncovered will help to develop a new era of precision wellness and nutrition.

Figure 4. Schematic representation of B-vitamin-mediated interaction between commensal leaner and host immunity.
Author Contributions
KY and KH wrote the draft of review article which was corrected by JK. KY, KH, and KS drew figures and JK performed correction.
Conflict of Involvement Statement
KS was employed past Nippon Flour Mills Co., Ltd.
The remaining authors declare that the research was conducted in the absenteeism of whatever commercial or financial relationships that could exist construed as a potential conflict of involvement.
Acknowledgments
This review article contains results obtained from our studies that were supported at least in role by grants from the Nihon Agency for Medical Research and Development [AMED; 17fk0108223h0002 (JK), 17ek0410032s0102 (JK), 17fk0108207h0002 (JK), 17ek0210078h0002 (JK), 17ak0101068h0001 (JK), 17gm1010006s0101 (JK), and 19ek0410062h0001 (JK)]; Cross-ministerial Strategic Innovation Promotion Program; the Ministry of Health, Labor, and Welfare of Nippon; the Science and Engineering science Inquiry Promotion Program for Agriculture, Forestry, Fisheries, and Food Industry; the Ministry of Educational activity, Culture, Sports, Science, and Engineering of Nihon; the Nippon Society for the Promotion of Science [JSPS, JP16H01373 (JK), JP15H05790 (JK), JP17H04134 (JK), JP26670241 (JK), JP26293111 (JK), JP18K17997 (KH), and JP18J00556 (KH)]; the ONO Medical Research Foundation (JK); the Catechism Foundation (JK); the Terumo Foundation for the Life Sciences and Arts; and the Nippon Ham Foundation for the Future of Food. KH is a JSPS Research Fellow.
References
4. Kau AL, Ahern PP, Griffin NW, Goodman AL, Gordon JI. Human diet, the gut microbiome, and immune organisation: envisioning the future. Nature. (2011) 474:327–36. doi: 10.1038/nature10213
CrossRef Total Text | Google Scholar
v. Lamichhane A, Kiyono H, Kunisawa J. Nutritional components regulate the gut allowed system and its association with intestinal immune disease evolution. J Gastroenterol Hepatol. (2013) 28(Suppl. four):xviii–24. doi: 10.1111/jgh.12259
PubMed Abstract | CrossRef Full Text | Google Scholar
vi. Whatham A, Bartlett H, Eperjesi F, Blumenthal C, Allen J, Suttle C, et al. Vitamin and mineral deficiencies in the developed world and their effect on the middle and vision. Ophthalmic Physiol Opt. (2008) 28:one–12. doi: 10.1111/j.1475-1313.2007.00531.x
PubMed Abstract | CrossRef Full Text | Google Scholar
10. Magnúsdóttir South, Ravcheev D, de Crécy-Lagard Five, Thiele I. Systematic genome assessment of B-vitamin biosynthesis suggests co-functioning among gut microbes. Front end Genet. (2015) 6:148. doi: 10.3389/fgene.2015.00148
PubMed Abstruse | CrossRef Full Text | Google Scholar
12. Hosomi Thousand, Kunisawa J. The specific roles of vitamins in the regulation of immunosurveillance and maintenance of immunologic homeostasis in the gut. Allowed Netw. (2017) 17:13–9. doi: 10.4110/in.2017.17.i.13
PubMed Abstract | CrossRef Full Text | Google Scholar
13. Suzuki H, Kunisawa J. Vitamin-mediated immune regulation in the evolution of inflammatory diseases. Endocr Metab Immune Disord Drug Targets. (2015) fifteen:212–5. doi: 10.2174/1871530315666150316122128
PubMed Abstract | CrossRef Full Text | Google Scholar
16. WHO/FAO. Vitamin and Mineral Requirements in Man Nutrition. 2nd ed. Report of a joint WHO/FAO Expert Consultation, Bangkok, Thailand 1998. Geneva: World Wellness Organization/Rome: Food and Agriculture Organization of the Un (2004).
twenty. Shikina T, Hiroi T, Iwatani K, Jang MH, Fukuyama South, Tamura M, et al. IgA course switch occurs in the organized nasopharynx- and gut-associated lymphoid tissue, but not in the diffuse lamina propria of airways and gut. J Immunol. (2004) 172:6259–64. doi: ten.4049/jimmunol.172.10.6259
CrossRef Full Text | Google Scholar
21. Kunisawa J, Sugiura Y, Wake T, Nagatake T, Suzuki H, Nagasawa R, et al. Style of bioenergetic metabolism during B cell differentiation in the intestine determines the distinct requirement for vitamin B1. Jail cell Rep. (2015) 13:122–31. doi: x.1016/j.celrep.2015.08.063
PubMed Abstruse | CrossRef Full Text | Google Scholar
22. Rindi M, Laforenza U. Thiamine intestinal send and related problems: contempo aspects. Proc Soc Exp Biol Med Soc Exp Biol Med. (2000) 224:246–55. doi: 10.1046/j.1525-1373.2000.22428.10
PubMed Abstruse | CrossRef Full Text | Google Scholar
23. Said HM. Recent advances in transport of h2o-soluble vitamins in organs of the digestive arrangement: a focus on the colon and the pancreas. Am J Physiol Gastrointest Liver Physiol. (2013) 305:G601–10. doi: x.1152/ajpgi.00231.2013
PubMed Abstruse | CrossRef Total Text | Google Scholar
24. Barrow BJ, Ortiz-Reyes R, O'Riordan MA, Pretlow TP. In situ localization of enzymes and mucin in normal rat colon embedded in plastic. Histochem J. (1989) 21:289–95. doi: 10.1007/BF01757182
PubMed Abstruse | CrossRef Full Text | Google Scholar
25. Nabokina SM, Inoue K, Subramanian VS, Valle JE, Yuasa H, Said HM. Molecular identification and functional characterization of the man colonic thiamine pyrophosphate transporter. J Biol Chem. (2014) 289:4405–sixteen. doi: 10.1074/jbc.M113.528257
PubMed Abstruse | CrossRef Full Text | Google Scholar
26. Lindhurst MJ, Fiermonte One thousand, Song Southward, Struys E, De Leonardis F, Schwartzberg PL, et al. Knockout of Slc25a19 causes mitochondrial thiamine pyrophosphate depletion, embryonic lethality, CNS malformations, and anemia. Proc Natl Acad Sci Us. (2006) 103:15927–32. doi: 10.1073/pnas.0607661103
PubMed Abstract | CrossRef Full Text | Google Scholar
27. Jurgenson CT, Begley TP, Ealick SE. The structural and biochemical foundations of thiamin biosynthesis. Annu Rev Biochem. (2009) 78:569–603. doi: 10.1146/annurev.biochem.78.072407.102340
PubMed Abstract | CrossRef Full Text | Google Scholar
28. Leonardi R, Roach PL. Thiamine Biosynthesis in Escherichia coli in vitro reconstruction of the thiazole synthase activity. J Biol Chem. (2004) 279:17054–62. doi: 10.1074/jbc.M312714200
CrossRef Full Text | Google Scholar
29. McRose D, Guo J, Monier A, Sudek S, Wilken S, Yan South, et al. Alternatives to vitamin B1 uptake revealed with discovery of riboswitches in multiple marine eukaryotic lineages. ISME J. (2014) eight:2517–29. doi: ten.1038/ismej.2014.146
PubMed Abstract | CrossRef Full Text | Google Scholar
30. Taylor SV, Kelleher NL, Kinsland C, Chiu HJ, Costello CA, Backstrom AD, et al. Thiamin biosynthesis in Escherichia coli. Identification of ThiS thiocarboxylate as the immediate sulfur donor in the thiazole formation J Biol Chem. (1998) 273:16555–threescore. doi: 10.1074/jbc.273.26.16555
PubMed Abstract | CrossRef Full Text | Google Scholar
31. Drywien Thou, Frackiewicz J, Górnicka Thou, Gadek J, Jałosinska M. Effect of probiotic and storage time of thiamine and riboflavin content in the milk drinks fermented by Lactobacillus casei KNE-1. Rocz Panstw Zakl Hig. (2015) 66:373–7.
PubMed Abstract | Google Scholar
32. Deguchi Y, Morishita T, Mutai M. Comparative studies on synthesis of water-soluble vitamins among human species of Bifidobacteria. Agric Biol Chem. (1985) 49:13–9.
Google Scholar
33. Masuda Thousand, Ide Thousand, Utsumi H, Niiro T, Shimamura Y, Murata M. Production potency of folate, vitamin B12, and thiamine by lactic acid bacteria isolated from Japanese pickles. Biosci Biotechnol Biochem. (2012) 76:2061–7. doi: 10.1271/bbb.120414
CrossRef Total Text | Google Scholar
34. Levit R, Savoy de Giori 1000, de Moreno de LeBlanc A, LeBlanc JG. Evaluation of the upshot of soymilk fermented by a riboflavin-producing Lactobacillus plantarum strain in a murine model of colitis. Benef Microbes. (2016) 8:65–72. doi: 10.3920/BM2016.0063
PubMed Abstract | CrossRef Total Text | Google Scholar
35. Juarez Del Valle M, Lai-o JE, de Moreno de LeBlanc A, Savoy de Giori Yard, LeBlanc JG. Soyamilk fermented with riboflavin-producing Lactobacillus plantarum CRL 2130 reverts and prevents ariboflavinosis in murine models. Br J Nutr. (2016) 116:1229–35. doi: 10.1017/S0007114516003378
PubMed Abstract | CrossRef Full Text | Google Scholar
36. Russo P, Capozzi Five, Arena MP, Spadaccino K, Due-as MT, López P, et al. Riboflavin-overproducing strains of Lactobacillus fermentum for riboflavin-enriched bread. Appl Microbiol Biotechnol. (2014) 98:3691–700. doi: ten.1007/s00253-013-5484-7
PubMed Abstract | CrossRef Full Text | Google Scholar
37. Shah Due north, Patel A. Recent advances in biosynthesis of vitamin and enzyme from nutrient grade leaner. Intl J Nutrient Ferment Technol. (2014) 4:79–85. doi: 10.5958/2321-7111.2015.00001.3
CrossRef Total Text | Google Scholar
39. Homayouni Rad A, Yari Khosroushahi A, Khalili M, Jafarzadeh Due south. Folate bio-fortification of yoghurt and fermented milk: a review. Dairy Sci Technol. (2016) 96:427–41. doi: 10.1007/s13594-016-0286-1
CrossRef Full Text | Google Scholar
40. Crittenden RG, Martinez NR, Playne MJ. Synthesis and utilisation of folate past yoghurt starter cultures and probiotic bacteria. Int J Nutrient Microbiol. (2003) 80:217–22. doi: x.1016/S0168-1605(02)00170-8
PubMed Abstruse | CrossRef Full Text | Google Scholar
41. D'Aimmo MR, Mattarelli P, Biavati B, Carlsson NG, Andlid T. The potential of bifidobacteria as a source of natural folate. J Appl Microbiol. (2012) 112:975–84. doi: x.1111/j.1365-2672.2012.05261.x
PubMed Abstract | CrossRef Full Text | Google Scholar
42. Song H, Yoo Y, Hwang J, Na Y-C, Kim HS. Faecalibacterium prausnitzii subspecies-level dysbiosis in the human gut microbiome underlying atopic dermatitis. J Allergy Clin Immunol. (2016) 137:852–60. doi: x.1016/j.jaci.2015.08.021
PubMed Abstract | CrossRef Full Text | Google Scholar
43. Piwowarek Yard, Lipinska E, Hać-Szymanczuk E, Kieliszek G, Scibisz I. Propionibacterium spp.-source of propionic acid, vitamin B12, and other metabolites important for the industry. Appl Microbiol Biotechnol. (2018) 102:515–38. doi: 10.1007/s00253-017-8616-7
PubMed Abstract | CrossRef Total Text | Google Scholar
44. Gu Q, Zhang C, Song D, Li P, Zhu 10. Enhancing vitamin B12 content in soy-yogurt past Lactobacillus reuteri. Int J Food Microbiol. (2015) 206:56–nine. doi: ten.1016/j.ijfoodmicro.2015.04.033
PubMed Abstract | CrossRef Full Text | Google Scholar
46. Costliow ZA, Degnan PH. Thiamine acquisition strategies bear upon metabolism and competition in the gut microbe Bacteroides thetaiotaomicron. mSystems. (2017) 2:17. doi: x.1128/mSystems.00116-17
PubMed Abstruse | CrossRef Full Text | Google Scholar
47. Almeida 50, Lochner M, Berod Fifty, Sparwasser T. Metabolic pathways in T cell activation and lineage differentiation. Semin Immunol. (2016) 28:514–24. doi: x.1016/j.smim.2016.10.009
PubMed Abstract | CrossRef Full Text | Google Scholar
48. Schramm 1000, Wiegmann Grand, Schramm S, Gluschko A, Herb M, Utermöhlen O, et al. Riboflavin (vitamin B2) deficiency impairs NADPH oxidase ii (Nox2) priming and defence force against Listeria monocytogenes. Eur J Immunol. (2014) 44:728–41. doi: ten.1002/eji.201343940
PubMed Abstract | CrossRef Total Text | Google Scholar
50. Sundaram U. Regulation of intestinal vitamin B2 absorption. Focus on "Riboflavin uptake past human being-derived colonic epithelial NCM460 cells." Am J Physiol Cell Physiol. (2000) 278:C268–9. doi: 10.1152/ajpcell.2000.278.2.C268
CrossRef Total Text | Google Scholar
51. Jaeger B, Bosch AM. Clinical presentation and outcome of riboflavin transporter deficiency: mini review later on five years of feel. J Inherit Metab Dis. (2016) 39:559–64. doi: x.1007/s10545-016-9924-two
PubMed Abstract | CrossRef Full Text | Google Scholar
52. Subramanian VS, Lambrecht N, Lytle C, Said HM. Conditional (intestinal-specific) knockout of the riboflavin transporter-3 (RFVT-iii) impairs riboflavin absorption. Am J Physiol - Gastrointest Liver Physiol. (2016) 310:G285–93. doi: 10.1152/ajpgi.00340.2015
PubMed Abstract | CrossRef Full Text | Google Scholar
53. Yonezawa A, Masuda S, Katsura T, Inui K. Identification and functional characterization of a novel man and rat riboflavin transporter, RFT1. Am J Physiol Cell Physiol. (2008) 295:C632–641. doi: x.1152/ajpcell.00019.2008
PubMed Abstract | CrossRef Total Text | Google Scholar
55. Tastan C, Karhan E, Zhou West, Fleming Due east, Voigt AY, Yao X, et al. Tuning of human MAIT cell activation past commensal bacteria species and MR1-dependent T-cell presentation. Mucosal Immunol. (2018) 11:1591–605. doi: 10.1038/s41385-018-0072-ten
PubMed Abstract | CrossRef Full Text | Google Scholar
56. Duurkens RH, Tol MB, Geertsma ER, Permentier HP, Slotboom DJ. Flavin binding to the high affinity riboflavin transporter RibU. J Biol Chem. (2007) 282:10380–six. doi: 10.1074/jbc.M608583200
PubMed Abstract | CrossRef Full Text | Google Scholar
57. Chen Z, Wang H, D'Souza C, Sun South, Kostenko Fifty, Eckle SBG, et al. Mucosal-associated invariant T-jail cell activation and aggregating after in vivo infection depends on microbial riboflavin synthesis and co-stimulatory signals. Mucosal Immunol. (2017) ten:58–68. doi: x.1038/mi.2016.39
PubMed Abstract | CrossRef Full Text | Google Scholar
58. Gold MC, Cerri S, Smyk-Pearson S, Cansler ME, Vogt TM, Delepine J, et al. Human mucosal associated invariant T cells find bacterially infected cells. PLoS Biol. (2010) 8:e1000407. doi: x.1371/journal.pbio.1000407
PubMed Abstract | CrossRef Full Text | Google Scholar
60. Le Bourhis 50, Dusseaux K, Bohineust A, Bessoles South, Martin E, Premel 5, et al. MAIT cells discover and efficiently lyse bacterially-infected epithelial cells. PLoS Pathog. (2013) ix:e1003681. doi: x.1371/journal.ppat.1003681
PubMed Abstract | CrossRef Full Text | Google Scholar
62. Kjer-Nielsen L, Patel O, Corbett AJ, Le Nours J, Meehan B, Liu Fifty, et al. MR1 presents microbial vitamin B metabolites to MAIT cells. Nature. (2012) 491:717–23. doi: 10.1038/nature11605
PubMed Abstract | CrossRef Full Text | Google Scholar
63. Serriari North-E, Eoche M, Lamotte L, Lion J, Fumery M, Marcelo P, et al. Innate mucosal-associated invariant T (MAIT) cells are activated in inflammatory bowel diseases. Clin Exp Immunol. (2014) 176:266–74. doi: 10.1111/cei.12277
PubMed Abstruse | CrossRef Total Text | Google Scholar
65. Singh N, Gurav A, Sivaprakasam Southward, Brady E, Padia R, Shi H, et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Amnesty. (2014) 40:128–39. doi: 10.1016/j.immuni.2013.12.007
PubMed Abstruse | CrossRef Full Text | Google Scholar
66. Lipszyc PS, Cremaschi GA, Zubilete MZ, Bertolino MLA, Capani F, Genaro AM, et al. Niacin modulates pro-inflammatory cytokine secretion. A potential mechanism involved in its anti-atherosclerotic effect Open Cardiovasc Med J. (2013) vii:90–viii. doi: 10.2174/1874192401307010090
PubMed Abstruse | CrossRef Full Text | Google Scholar
67. Amanullah S, Seeber C. Niacin deficiency resulting in neuropsychiatric symptoms: A case study and review of literature. Clin Neuropsychiatry. (2010) vii:10–xiv.
Google Scholar
68. Gao J, Xu K, Liu H, Liu G, Bai Chiliad, Peng C, et al. Touch of the gut microbiota on intestinal immunity mediated by tryptophan metabolism. Forepart Cell Infect Microbiol. (2018) 8:13. doi: 10.3389/fcimb.2018.00013
PubMed Abstruse | CrossRef Full Text | Google Scholar
69. Gazzaniga F, Stebbins R, Chang SZ, McPeek MA, Brenner C. Microbial NAD metabolism: lessons from comparative genomics. Microbiol Mol Biol Rev. (2009) 73:529–41. doi: ten.1128/MMBR.00042-08
PubMed Abstruse | CrossRef Total Text | Google Scholar
70. Kurnasov O, Goral V, Colabroy K, Gerdes South, Anantha S, Osterman A, et al. NAD biosynthesis: identification of the tryptophan to quinolinate pathway in leaner. Chem Biol. (2003) x:1195–204. doi: 10.1016/j.chembiol.2003.eleven.011
PubMed Abstruse | CrossRef Full Text | Google Scholar
71. Rodionov DA, Li Ten, Rodionova IA, Yang C, Sorci L, Dervyn Due east, et al. Transcriptional regulation of NAD metabolism in bacteria: genomic reconstruction of NiaR (YrxA) regulon. Nucleic Acids Res. (2008) 36:2032–46. doi: 10.1093/nar/gkn046
PubMed Abstruse | CrossRef Full Text | Google Scholar
73. Hodges RE, Ohlson MA, Bean WB. Pantothenic acid deficiency in man1. J Clin Invest. (1958) 37:1642–57. doi: 10.1172/JCI103756
CrossRef Total Text | Google Scholar
74. Yang M, Moclair B, Hatcher V, Kaminetsky J, Mekas Thousand, Chapas A, et al. A randomized, double-bullheaded, placebo-controlled written report of a novel pantothenic Acid-based dietary supplement in subjects with balmy to moderate facial acne. Dermatol Ther. (2014) 4:93–101. doi: 10.1007/s13555-014-0052-three
PubMed Abstruse | CrossRef Full Text | Google Scholar
75. Kobayashi D, Kusama M, Onda M, Nakahata Northward. The upshot of pantothenic acid deficiency on keratinocyte proliferation and the synthesis of keratinocyte growth factor and collagen in fibroblasts. J Pharmacol Sci. (2011) 115:230–4. doi: 10.1254/jphs.10224SC
PubMed Abstract | CrossRef Full Text | Google Scholar
77. Zhang B, Lo C, Shen L, Sood R, Jones C, Cusmano-Ozog K, et al. The role of vanin-one and oxidative stress–related pathways in distinguishing acute and chronic pediatric ITP. Blood. (2011) 117:4569–79. doi: 10.1182/blood-2010-09-304931
PubMed Abstract | CrossRef Full Text | Google Scholar
78. Berruyer C, Pouyet L, Millet V, Martin FM, LeGoffic A, Canonici A, et al. Vanin-1 licenses inflammatory mediator production by gut epithelial cells and controls colitis by antagonizing peroxisome proliferator-activated receptor gamma activity. J Exp Med. (2006) 203:2817–27. doi: 10.1084/jem.20061640
PubMed Abstract | CrossRef Full Text | Google Scholar
79. He W, Hu Due south, Du X, Wen Q, Zhong X-P, Zhou X, et al. Vitamin B5 reduces bacterial growth via regulating innate immunity and adaptive immunity in mice infected with Mycobacterium tuberculosis. Front Immunol. (2018) 9:365. doi: x.3389/fimmu.2018.00365
PubMed Abstract | CrossRef Full Text | Google Scholar
81. Parra M, Stahl Southward, Hellmann H. Vitamin B6 and its function in jail cell metabolism and physiology. Cells. (2018) vii:84. doi: x.3390/cells7070084
CrossRef Full Text | Google Scholar
83. Huang South-C, Wei JC-C, Wu DJ, Huang Y-C. Vitamin B (half-dozen) supplementation improves pro-inflammatory responses in patients with rheumatoid arthritis. Eur J Clin Nutr. (2010) 64:1007–thirteen. doi: 10.1038/ejcn.2010.107
PubMed Abstract | CrossRef Full Text | Google Scholar
84. Jannusch K, Jockwitz C, Bidmon H-J, Moebus S, Amunts K, Caspers Due south. A circuitous interplay of vitamin B1 and B6 metabolism with knowledge, brain structure, and functional connectivity in older adults. Front Neurosci. (2017) eleven:596. doi: 10.3389/fnins.2017.00596
PubMed Abstruse | CrossRef Full Text | Google Scholar
85. Qian B, Shen S, Zhang J, Jing P. Effects of vitamin B6 deficiency on the composition and functional potential of T cell populations. J Immunol Res. (2017) 2017:2197975. doi: ten.1155/2017/2197975
PubMed Abstract | CrossRef Full Text | Google Scholar
86. Chiang E-PI, Selhub J, Bagley PJ, Dallal G, Roubenoff R. Pyridoxine supplementation corrects vitamin B6 deficiency but does not better inflammation in patients with rheumatoid arthritis. Arthritis Res Ther. (2005) 7:R1404–eleven. doi: 10.1186/ar1839
CrossRef Full Text | Google Scholar
xc. Ikeda M, Miyamoto A, Mutoh S, Kitano Y, Tajima One thousand, Shirakura D, et al. Development of biotin prototrophic and hyper-auxotrophic Corynebacterium glutamicum strains toward biotin product. Appl Env Microbiol. (2013) 2013:828. doi: 10.1128/AEM.00828-13
CrossRef Full Text | Google Scholar
91. Ikeda M, Nagashima T, Nakamura E, Kato R, Ohshita Chiliad, Hayashi Thousand, et al. In vivo roles of fat acid biosynthesis enzymes in biosynthesis of biotin and α-lipoic acid in Corynebacterium glutamicum. Appl Environ Microbiol. (2017) 83:17. doi: 10.1128/AEM.01322-17
PubMed Abstract | CrossRef Full Text | Google Scholar
92. Kothapalli North, Camporeale G, Kueh A, Chew YC, Oommen AM, Griffin JB, et al. Biological functions of biotinylated histones. J Nutr Biochem. (2005) 16:446–8. doi: ten.1016/j.jnutbio.2005.03.025
PubMed Abstruse | CrossRef Full Text | Google Scholar
95. Agrawal S, Agrawal A, Said HM. Biotin deficiency enhances the inflammatory response of human dendritic cells. Am J Physiol Cell Physiol. (2016) 311:C386–391. doi: ten.1152/ajpcell.00141.2016
PubMed Abstract | CrossRef Full Text | Google Scholar
96. Elahi A, Sabui S, Narasappa NN, Agrawal S, Lambrecht NW, Agrawal A, et al. Biotin deficiency induces Th1- and Th17-mediated proinflammatory responses in human CD4+ T lymphocytes via activation of the mTOR signaling pathway. J Immunol Baltim Doctor. (2018) 200:2563–70. doi: 10.4049/jimmunol.1701200
PubMed Abstract | CrossRef Full Text | Google Scholar
100. Entcheva P, Phillips DA, Streit WR. Functional analysis of Sinorhizobium meliloti genes involved in biotin synthesis and transport. Appl Environ Microbiol. (2002) 68:2843–8. doi: 10.1128/AEM.68.6.2843-2848.2002
PubMed Abstract | CrossRef Full Text | Google Scholar
101. Sabui South, Bohl JA, Kapadia R, Cogburn K, Ghosal A, Lambrecht NW, et al. Role of the sodium-dependent multivitamin transporter (SMVT) in the maintenance of intestinal mucosal integrity. Am J Physiol Gastrointest Liver Physiol. (2016) 311:G561–lxx. doi: ten.1152/ajpgi.00240.2016
PubMed Abstruse | CrossRef Total Text | Google Scholar
102. Fisher DJ, Fernández RE, Adams NE, Maurelli AT. Uptake of biotin past Chlamydia Spp. through the use of a bacterial transporter (BioY) and a host-prison cell transporter (SMVT). PLoS Ane. (2012) vii:e46052. doi: ten.1371/periodical.pone.0046052
PubMed Abstruse | CrossRef Full Text | Google Scholar
103. Hayashi A, Mikami Y, Miyamoto Thousand, Kamada N, Sato T, Mizuno South, et al. Intestinal dysbiosis and biotin deprivation induce alopecia through overgrowth of Lactobacillus murinus in mice. Cell Rep. (2017) 20:1513–24. doi: 10.1016/j.celrep.2017.07.057
PubMed Abstract | CrossRef Full Text | Google Scholar
104. Sugahara H, Odamaki T, Fukuda S, Kato T, Xiao J, Abe F, et al. Probiotic Bifidobacterium longum alters gut luminal metabolism through modification of the gut microbial customs. Sci Rep. (2015) v:13548. doi: 10.1038/srep13548
PubMed Abstract | CrossRef Full Text | Google Scholar
105. Courtemanche C, Elson-Schwab I, Mashiyama ST, Kerry Due north, Ames BN. Folate deficiency inhibits the proliferation of master human CD8+ T lymphocytes in vitro. J Immunol Baltim Md. (2004) 173:3186–92. doi: x.4049/jimmunol.173.five.3186
PubMed Abstract | CrossRef Full Text | Google Scholar
106. Yamaguchi T, Hirota K, Nagahama Chiliad, Ohkawa K, Takahashi T, Nomura T, et al. Command of allowed responses past antigen-specific regulatory T cells expressing the folate receptor. Amnesty. (2007) 27:145–59. doi: 10.1016/j.immuni.2007.04.017
PubMed Abstruse | CrossRef Full Text | Google Scholar
107. Kinoshita M, Kayama H, Kusu T, Yamaguchi T, Kunisawa J, Kiyono H, et al. Dietary folic acid promotes survival of Foxp3+ regulatory T cells in the colon. J Immunol. (2012) 189:2869–78. doi: x.4049/jimmunol.1200420
PubMed Abstruse | CrossRef Total Text | Google Scholar
108. Kunisawa J, Hashimoto E, Ishikawa I, Kiyono H. A pivotal role of vitamin B9 in the maintenance of regulatory T cells in vitro and in vivo. PLoS ONE. (2012) vii:e32094. doi: 10.1371/periodical.pone.0032094
PubMed Abstract | CrossRef Full Text | Google Scholar
109. Sakaguchi Southward, Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T. Regulatory T cells: how do they suppress immune responses? Int Immunol. (2009) 21:1105–xi. doi: 10.1093/intimm/dxp095
PubMed Abstract | CrossRef Total Text | Google Scholar
110. Melse-Boonstra A, de Bree A, Verhoef P, Bjørke-Monsen AL, Verschuren WMM. Dietary monoglutamate and polyglutamate folate are associated with plasma folate concentrations in Dutch men and women aged xx-65 years. J Nutr. (2002) 132:1307–12. doi: x.1093/jn/132.6.1307
PubMed Abstract | CrossRef Total Text | Google Scholar
111. Zhao R, Diop-Bove North, Visentin M, Goldman ID. Mechanisms of membrane send of folates into cells and across epithelia. Annu Rev Nutr. (2011) 31:177–201. doi: 10.1146/annurev-nutr-072610-145133
PubMed Abstract | CrossRef Total Text | Google Scholar
112. Maguire F, Henriquez F L, Leonard Grand, Dacks JB, Chocolate-brown MW, Richards TA. Circuitous patterns of gene fission in the eukaryotic folate biosynthesis pathway. Genome Biol Evol. (2014) vi:2709–20. doi: ten.1093/gbe/evu213
PubMed Abstruse | CrossRef Total Text | Google Scholar
114. Strozzi GP, Mogna L. Quantification of folic acid in human feces later administration of Bifidobacterium probiotic strains. J Clin Gastroenterol. (2008) 42(Suppl. 3):S179–84. doi: 10.1097/MCG.0b013e31818087d8
PubMed Abstract | CrossRef Total Text | Google Scholar
115. Santos F, Wegkamp A, de Vos WM, Smid EJ, Hugenholtz J. Loftier-level folate production in fermented foods by the B12 producer Lactobacillus reuteri JCM1112. Appl Environ Microbiol. (2008) 74:3291–iv. doi: ten.1128/AEM.02719-07
PubMed Abstract | CrossRef Total Text | Google Scholar
116. Araújo MM, Marchioni Eastward, Villavicencio ALCH, Zhao M, di Pascoli T, Kuntz F, et al. Mechanism of folic acid radiolysis in aqueous solution. LWT - Food Sci Technol. (2015) 63:599–603. doi: 10.1016/j.lwt.2015.03.038
CrossRef Full Text | Google Scholar
117. Patel O, Kjer-Nielsen Fifty, Le Nours J, Eckle SBG, Birkinshaw R, Beddoe T, et al. Recognition of vitamin B metabolites by mucosal-associated invariant T cells. Nat Commun. (2013) 4:2142. doi: ten.1038/ncomms3142
PubMed Abstract | CrossRef Full Text | Google Scholar
118. Eckle SBG, Birkinshaw RW, Kostenko Fifty, Corbett AJ, McWilliam HEG, Reantragoon R, et al. A molecular basis underpinning the T jail cell receptor heterogeneity of mucosal-associated invariant T cells. J Exp Med. (2014) 211:1585–600. doi: 10.1084/jem.20140484
PubMed Abstract | CrossRef Full Text | Google Scholar
120. Tamura J, Kubota K, Murakami H, Sawamura M, Matsushima T, Tamura T, et al. Immunomodulation by vitamin B12: augmentation of CD8+ T lymphocytes and natural killer (NK) cell activeness in vitamin B12-deficient patients by methyl-B12 treatment. Clin Exp Immunol. (1999) 116:28–32. doi: 10.1046/j.1365-2249.1999.00870.x
PubMed Abstract | CrossRef Full Text | Google Scholar
121. Beedholm-Ebsen R, van de Wetering M, Hardlei T, Nexø E, Borst P, Moestrup SK. Identification of multidrug resistance protein 1 (MRP1/ABCC1) as a molecular gate for cellular export of cobalamin. Blood. (2010) 115:1632–9. doi: 10.1182/claret-2009-07-232587
PubMed Abstruse | CrossRef Total Text | Google Scholar
123. Patel A, Shah North, Prajapati JB. Biosynthesis of vitamins and enzymes in fermented foods by lactic acid bacteria and related genera - a promising arroyo. Croat J Nutrient Sci Technol. (2013) v:85–91.
Google Scholar
saultersmurst1985.blogspot.com
Source: https://www.frontiersin.org/articles/10.3389/fnut.2019.00048/full#:~:text=Thus%2C%20many%20intestinal%20bacteria%20from,are%20important%20for%20host%20immunity.
0 Response to "Which Vitamin Can Be Produced by Intestinal Bacteria?"
Post a Comment